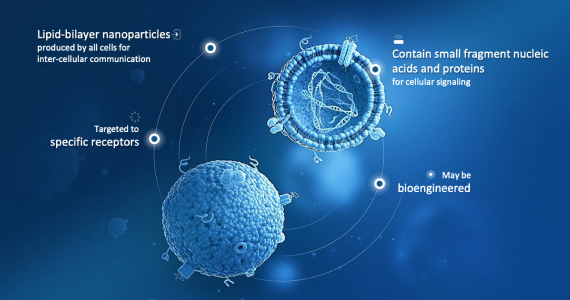
Capricor’s Exosomes Technology
StealthX™ is Capricor’s proprietary exosome platform for designer protein surface expression, cargo loading and targeted delivery. StealthX™ technology is at the core of our exosome platform, and we are currently exploring its deployment in two broad modalities: precision therapeutics and vaccinology.
The ability to decorate the surface of exosomes with proteins or other ligands or antigens, as well as to load potential therapeutic cargo inside the exosome coupled with their relatively low toxicity compared to synthetic lipid nanoparticles, allows us to explore a number of potential disease targets.
Capricor is internally developing a streamlined manufacturing process to support future development efforts. This foundational work drives our platform strategy to leverage the development of external partnerships with companies that have therapeutics that can benefit from our StealthX™ targeted delivery vehicle. Our StealthX™ platform builds on advances in fundamental protein and RNA science, targeting technology and biological manufacturing, providing Capricor the opportunity to unlock the potential power of exosomes as an exciting modality for drug delivery.
We are currently collaborating with the National Institutes of Health (NIH) investigating our proprietary StealthX™ exosome-based vaccine (StealthX™ vaccine) for the prevention of SARS-CoV-2. Our vaccine was selected to be part of Project NextGen, an initiative by the U.S. Department of Health and Human Services to advance a pipeline of new, innovative vaccines providing broader and more durable protection for COVID-19 and other infectious threats. As part of Project NextGen, the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health is conducting a Phase 1 clinical study with our StealthX™ vaccine. NIAID's Division of Microbiology and Infectious Diseases (DMID) is overseeing the study. For more information, click here.